Applied Clinical Trials (written by Moe Alsumidaie from AnnexClinical) just released a fascinating article detailing the staggering financial impact of participant non-adherence to study protocol in clinical trials.
It’s easy to overlook the financial ramifications to participant non-adherence. “Non-Adherence: A Direct Influence on Clinical Trial Duration and Cost” makes this information extremely accessible, using charts and graphs to emphasize these standout financial ramifications:
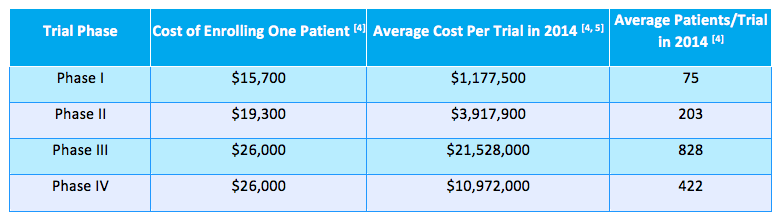
- The cost of enrollment for clinical trials is huge. Costs range from $15,700 per participant for a smaller phase 1 trial all the way up to $26,600 for larger phase 3 & 4 trials.
- 40% of patients become non-adherent to the investigational medical product by the 150 day mark of a clinical trial. This giant percentage results in skewed statistical results, not to mention the potential risks to the participant due to skipping doses or double-dosing. Medication administration is an area of non-adherence that affects trial results to a large degree.
- “Non-Adherence…” takes this information a step further by detailing just how many participants would need to be added to a trial to off-set the statistical variability produced by non-adherence. The result is hundreds of thousands, if not millions, of dollars lost on each clinical trial due to non-adherence.
Text Messaging Medication Reminders
Text messaging software offers significant solutions for the most common non-adherence issues in clinical trials. While impossible to completely eliminate, it IS possible to minimize non-adherence through text-based reminders, alerts, and interventions.
Medication Reminders
Text messaging enables you to send SMS medication administration reminders directly to a study participant’s mobile device without requiring that they use a smart phone or downloadable app. You can use this type of reminder to let them know when take their medication or use a device, timed out for your specific study’s needs. You can even ask participants to send a confirmatory reply using an alphanumerical character, commonly Y or N (yes or no) to confirm whether or not they are complying with study protocol. A medication reminder app would work similarly in alerting and reminding participants about specific dosing and timing instructions. Completion nudges can be sent when patients haven’t confirmed to ensure timely compliance.
Text-based Alerts and Interventions
Keeping participants engaged in complex study protocols, sometimes lasting for years, can be challenging. Sending timely alerts and reminders gives you another opportunity to keep participants on track.
Gather Additional Data via SMS Surveys
Along with Y/N confirmations, additional questions can be asked of patients via SMS Survey to collect follow-on data, such as dosing dates/times.
Conditional Triggers and Logic Applications
Based in input data, additional functions can take place within the study. For example, if a patient misses a dose, study staff can be notified so actions can be taken or a new series of reminders can be deployed to ensure the patient stays on schedule.
Utilize Photo Messaging to Confirm Compliance
Increasing site visits is often not convenient or cost effective. You can use this type of text to your advantage by aiding in the confirmation of study compliance. Ask participants to send photos of their stat-tracking journal, medications, dosing schedule, or even using a device if applicable to the study. Mobile devices can also be used to take photos of labels, products, etc.
Why Text Messaging Works Better than Anything Else
- 91% of mobile users keep their phone within arm’s reach nearly 24/7.
- 90% of text messages are read within the first 3 minutes of receipt.
- 23% of phone calls go straight to voicemail…30% of which go unheard for 3 days.
Text messaging is an extremely direct and efficient way to engage study patients.
The costs of clinical trial non-adherence are far from just financial, and the smallest increase in study participant adherence goes far. Text messaging, along with the utilization of a medication reminder app, can be a huge asset for clinical trials that rely on adherence to protocol.
For more information on how Mosio’s medication reminders via text messaging app, contact us or view pricing.