Clinical trials are extremely important to the development of new drugs and other medical devices, but they’re not without their challenges. With ever-increasing regulations and standards, clinical trials can be costly, time-consuming and, most importantly, they can put human lives at risk if they aren’t conducted correctly.
Wearables in clinical trials are making that process more efficient, effective and secure by improving monitoring of participants’ health as well as by giving researchers more data to work with than ever before so they can develop more effective treatments and drugs.
Why Use Wearables In Clinical Trials?
There are a number of reasons why clinical trial sponsors might consider incorporating wearable technology into their study design:
- Increase patient compliance
- Help improve data quality
- Save time
Below are some examples of clinical trials that incorporate wearable technology into their study design. Although these trials cover a wide range of therapeutic areas, there is one common thread: all studies use wearable technology to help collect more consistent and accurate data from participants.
- Johnson & Johnson Heartline study
- Parkinson’s Disease study
- Using Smart Watches to Detect and Monitor COVID-19
The one advantage that wearables have over other types of patient-reported outcome data is a direct connection to patient behavior. There are always going to be patients who don’t fill out their questionnaires or complete every task they’re assigned, and clinical trial sponsors will often run into problems when analyzing these kinds of data.
For example, if you ask a participant how many times they exercised during a certain period but they only exercise twice in that time frame, how do you know whether they were telling you the truth or if they are remembering correctly? With wearables, those questions are easy to answer.
There are several possible explanations for why patient behavior might differ from what is captured in questionnaires. One explanation is that patients might be feeling particularly good or bad on a given day, leading them to underestimate or overestimate their daily activities. Another reason could be that they didn’t take their pills or they aren’t following your protocol precisely as instructed. These kinds of issues can have a negative impact on data quality and lead to skewed results if not addressed.
Who Uses Wearables In Clinical Trials?
Besides patients, you’ll also find wearables being used in clinical trials by trial sites, medical devices companies and medical device researchers. Trial sites benefit from wearable technology because it offers new ways to interact with patients in an unobtrusive way. Essentially, it frees trial sites from sticking to the previous face-to-face forms of communication.
Medical device companies benefit from wearable technology because it increases their understanding of patient experience with their products. The more a company understands about how patients interact with its devices, for example, and whether patients like using them, then the more effectively it can improve those devices. To make informed design changes, medical device companies use data gathered by wearables to better understand how people use and respond to their products.
After gathering data, medical device companies typically use wearables data to improve their products in three ways:
- Identifying issues with their design
- Gaining insights into which users are struggling to use their devices
- Informing improvements to their interface
How Are Wearables Are Used to Get Additional Information
More advanced wearables are designed to be worn for extended periods of time and can collect information about things like heart rate, blood pressure, oxygen levels, activity levels, etc.
For example, a blood-pressure wearable could track blood pressure for an entire day (vs. 1-2 readings taken by a doctor during a visit). This provides more accurate data that can help patients with hypertension better manage their condition.
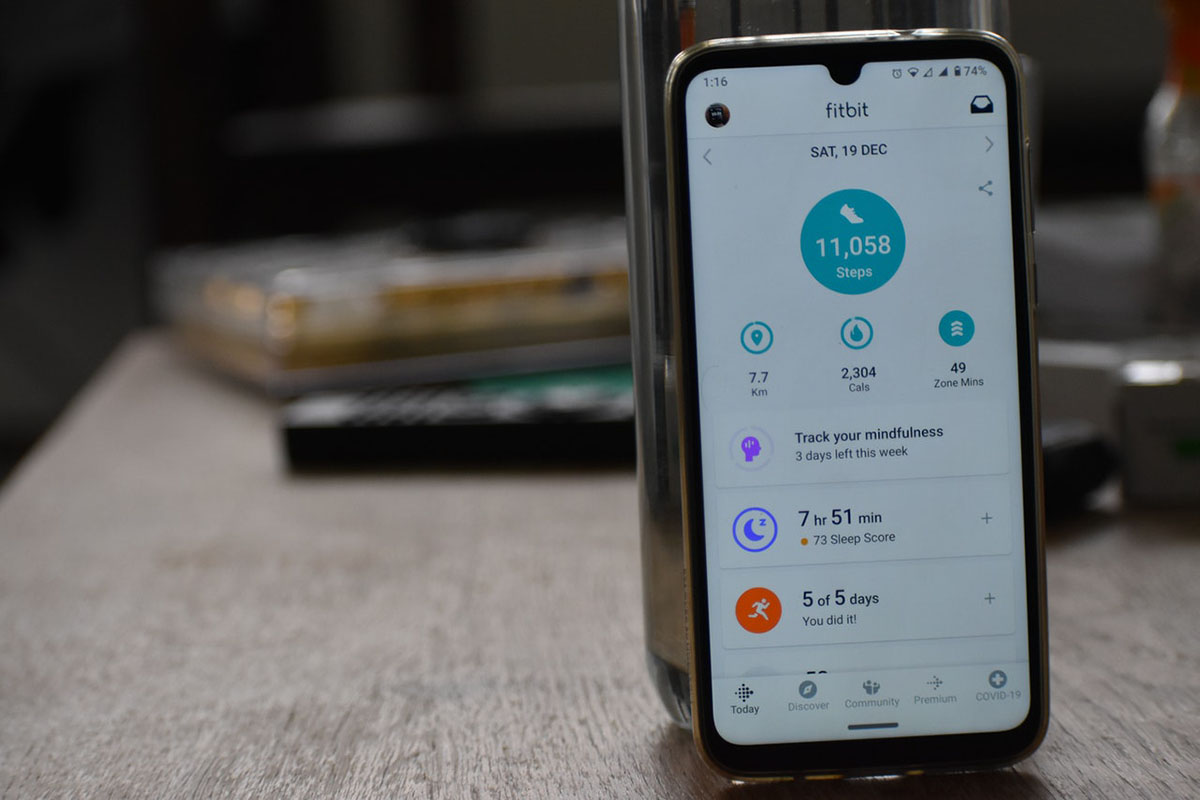
With many wearables, patients can easily access their data on demand through a smartphone app, which allows them to actively manage their own health and wellness. The real-time data collected by these devices also means that researchers will be able to identify problems early on and intervene before things get worse.
Additionally, wearable technology is significantly more comfortable than traditional monitoring methods like blood draws or glucose meters—many trackers are even waterproof! This makes it easier for patients to monitor themselves throughout each day and collect comprehensive health data over time since they don’t have to worry about the device getting wet from bathing, rain, or swimming.
Challenges Of Using And Conducting Studies On Wearables
There are also a number of challenges associated with using wearables in clinical trials. Patients need to be willing and able to use their devices as instructed. Some patients may not fully understand how to use a device, or may not have access to regular charging facilities if they’re wearing a tracker all day long.
Other patients might not want to be bothered with another device that requires data input—not everyone wants full-time access to their health data, especially if it requires them having a smartphone on hand all day!
Unlike traditional methods of gathering data about patients, such as blood samples and interviews, wearables are new technology. Because of their novelty, not all wearables have not been through stringent scientific testing in regards to their reliability or accuracy in measurement.
In addition to these concerns about wearable technologies’ validity in measuring clinical parameters for trials, there are also doubts about whether patients will actually use wearables willingly over long periods of time when monitored by a health professional.
One of the primary concerns surrounding wearable use in clinical trials is patient participation. If a patient doesn’t want to wear their device, or if they become uncomfortable with having their data tracked over long periods of time, then compliance will be hard to come by.
In order to help ensure that participants will continue wearing their devices and stay on track throughout a trial, most clinical trial specialists agree that patients should receive some sort of direct benefit for participating. This could take any form such as receiving medication at a reduced cost, gaining access to other services provided by health care professionals or getting early access to new medications not yet available on the market.
Another hurdle for clinicians is that wearables vary greatly in terms of quality, features, and cost. When it comes to selecting which wearable device will be best for a clinical trial, clinicians must first decide what they are looking for in a device.
A wearable that measures heart rate and provides wireless data transfer capabilities might be better suited to a cardiovascular study than one that measures blood pressure but can only sync wirelessly via computer. Clinicians must also consider how their patient population will react to using specific types of wearables as not everyone finds them appealing or even comfortable enough to want to use.
Clinicians should also take into account any regulations surrounding use of wearables in clinical trials as well as how to actually collect and store data that comes from wearable devices. Some aspects of a trial may be easily measured by wearables, but others may not be able to.
Once an appropriate wearable device has been selected for a study, then clinicians will have to figure out what specific measurements and data they want to track from it. Before tracking can begin, both patient and clinician must agree on exactly what is going to be tracked from both sides’ perspectives. This information will go into informed consent documents that are signed by both parties before participating in any study with a wearable device.
Benefits of Wearable Technology in Clinical Trials
There are several ways that wearable technology in clinical trials may be useful:
- Saves time and money
- Improves data quality
- Allows faster test results
- Increases patient participation
- Provides opportunities for secondary research
- Can help prevent side effects of drugs or treatments by identifying problems early on
One of the benefits of wearable technology in clinical trials is saving time and money. By using smartwatches, researchers can monitor and track patients’ heart rate and body temperature 24/7. This means they don’t have to bring patients into hospitals frequently to collect data that could be collected on a continuous basis by wearables.
Saving time is also possible because wearing these devices helps prevent delays that are often caused when manual testing is used to identify problems with drugs or treatments before clinical trials take place. These delays can cause lost time in finding solutions that may help improve drugs or treatments.
Moreover, not only does wearable technology reduce costs, but it also increases data quality because all data is automatically recorded, which reduces human error compared to manual recording methods.
Another benefit of wearable technology in clinical trials is that it allows faster test results. Smartwatches and other wearables can monitor patients’ vital signs, such as heart rate and body temperature, while they’re sleeping or when they have symptoms that suggest an adverse reaction to drugs or treatments.
This makes it easier for researchers to recognize adverse reactions that otherwise may be difficult to detect with traditional methods, which can then speed up changes in drugs or treatments before larger clinical trials are done.
Wearable technology provides an early warning system against problems with drugs or treatments before larger-scale studies are conducted. A faster response may help prevent injuries caused by a drug’s side effects, which in turn could save a lot of money and lives by improving public health.
Another benefit of wearable technology in clinical trials is that it increases patient participation. Research has shown that some participants enjoy wearing a smartwatch because it’s an extension of their phone and allows them to connect with friends, family and loved ones.
The ability to get texts and calls from family members may improve mental health in patients because those with depression are more likely to report feeling connected to others when texting rather than talking face-to-face, according to a study published in Emotion. Some wearables even include an optional pulse reader for patients who may have anxiety about using an ECG machine at every visit.
Additional opportunities for research are available to researchers since they will have vital information of patients who used the devices during the study. There may be new insights that can be gleaned from the additional data.
Vitals information can also be used as an early warning signal for any potential side effects. Researchers can monitor trends in a patient’s vitals to be on the lookout for potential issues.
Do Wearables Clinical Trials Need FDA Approval?
The short answer is no. However, wearables used in clinical trials do have to be approved by whoever manages your study. Wearables are low-risk technology because they don’t interact with or diagnose patients in any way, which makes them more akin to something like a scale than an actual medical device used in patient care—something that would typically require FDA approval before being sold to consumers.
The decision to use a wearable in a study is up to the study manager of the clinical trial. Researchers can help make the decision by determining whether or not a wearable device falls under one of three categories:
The first category—low-risk technology—covers items like scales and activity trackers that don’t interact with patients or diagnose illnesses. These devices are typically exempt from FDA approval if they’re being used for low-risk studies, such as a small side-study that’s meant to supplement data from a larger main study.
The second category—high-risk technology—covers medical devices and medical equipment that often requires special training to use correctly and could potentially cause harm if used incorrectly.
The third category—devices that fall somewhere in between—covers items like ECG monitors and portable pulse oximeters. Items that fall into this category have some risks, but not enough to require FDA approval. These devices can sometimes be used without further regulatory approval, but the study manager will ultimately decide whether or not they’re allowed.
Some features of smartwatches are getting FDA approval. Apple Watch’s heart rate monitoring feature was recently approved by FDA as a medical device, though only for consumer use and not for diagnosing or treating any medical conditions. FDA regulations also require that all data collected by wearable devices is sent back to home base via Bluetooth or Wifi link which may not always be possible in remote locations such as rural Africa where patient compliance may be an issue.

Clinical Trials Using Wearables Today
Many medical and healthcare professionals are investigating how wearable technology could track patient vitals, such as blood pressure or heart rate, in real time. Physicians may even be able to use wearables to remotely monitor patients’ progress after a procedure.
For example, researchers at Stanford University recently developed an algorithm that uses an Apple Watch to detect arrhythmias (irregular heartbeats). The device is currently being tested on people with implanted pacemakers. If successful, it could help save lives by providing a less invasive way of monitoring cardiac activity than traditional EKG tests.
This new wave of technology allows researchers to use wearables in different ways and collect more data than ever before while improving patient comfort and privacy. It could also bring down costs by reducing how often patients must visit their physician or need additional tests. Ultimately, wearable technology provides a powerful new way for researchers to monitor patient health in real time with minimal interference and provide instant feedback based on that data.
Wearable Technologies for Clinical Trials
Activity Trackers
- Fitbit
- Garmin
Smartwatches
- Apple Watch
- Samsung Gear
- Pebble
Optical & Bio-Based Sensors
- Scioly
- us
Vitals
- Biotronik Lifestar
- Apple Watch
Moisture Sensors
- SiProX – Waterproof IP67 Sensor Monitoring
At present, wearable technology is still an emerging concept and has only been adopted in a limited number of clinical trials. However, there are multiple examples where wearables have shown promising results. Researchers at Cedars-Sinai Medical Center used Fitbits to monitor the functional status cancer patients were getting. Aetna Insurance started a program using Apple Watch to track blood pressure and reward patients’ healthy behaviors.
By using these technologies, physicians can keep a close eye on their patient’s well-being and take timely actions if required. Even though it is still early days for wearable technology in clinical trials it holds great potential. Deciding whether or not to use wearables as part of your study should be done as a group, all benefits and challenges should be discussed as part of the decision.








